 
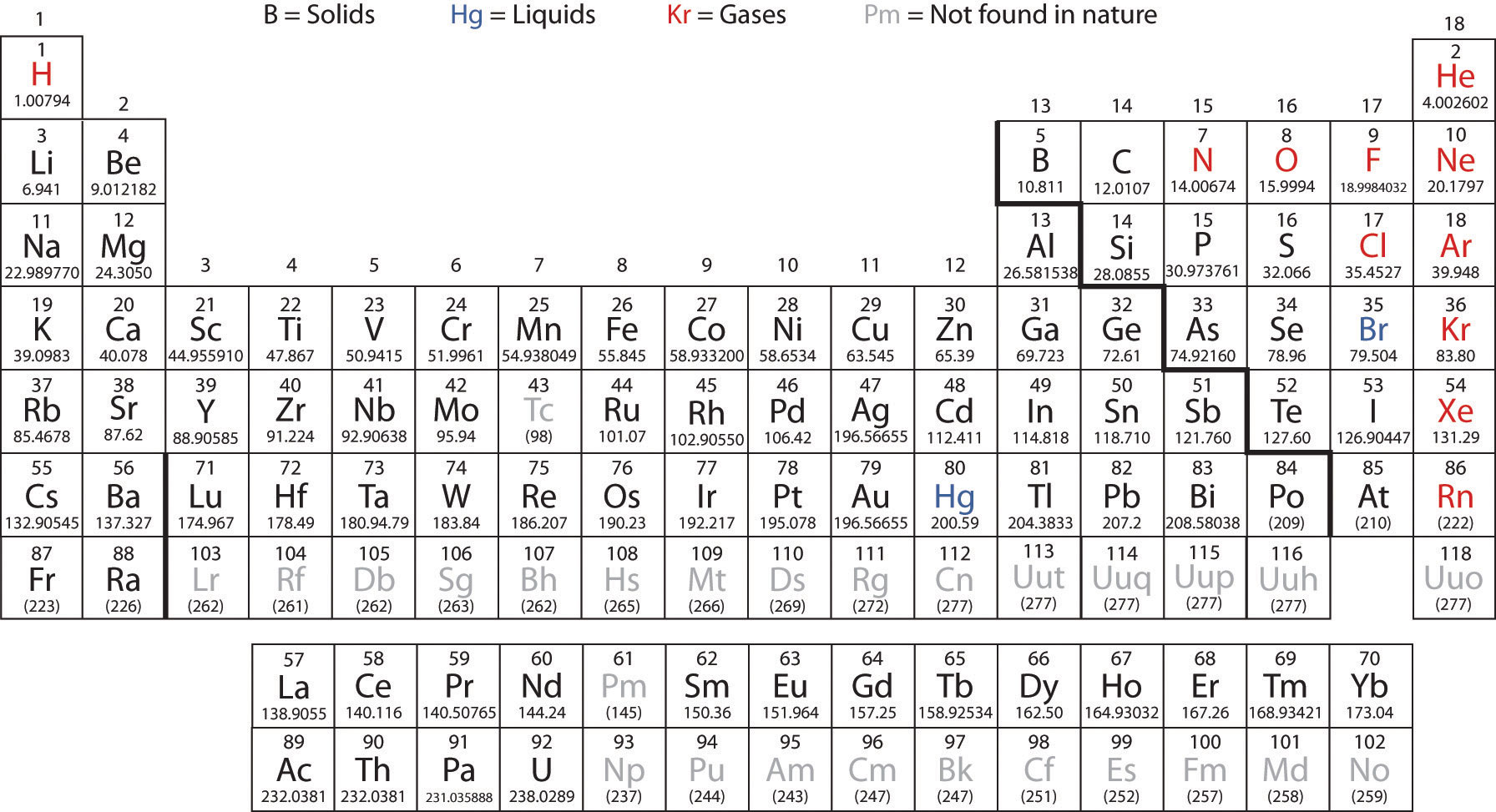
These are similar to Bohr Diagrams, but only provide information about the valence electrons (the outer electrons of an element). We talked in quite some depth both about the general properties of these groups and also about some of the properties of the elements themselves: We also talked about some of the groups of the Periodic Table – alkali metals, alkaline Earth metals, halogens and noble gases. We went on to talk about some of the information in the Periodic Table such as atomic number, atomic weight, chemical symbols & the element names They also did the worksheet I made for them which has them look at the number of shells and number of valence electrons various elements have. We used this chart a lot as we continued on with this unit because then they could easily see how many valence electrons were in the outer shell of the elements! The next day, the kids did a cut-and-paste activity with those same Bohr Diagrams. Note: There are Bohr Diagram cards for elements 1 (hydrogen) through 20 (calcium) included in the Chemistry Packet.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
